Atomic theory timeline4/16/2024  Consequently a given compound always has the same relative number and types of atoms. Thus, in a pure compound atoms of unlike elements would be combined in small whole number ratios. If one assumed that elements were made of the same type of indivisible particles, that are identical in mass and all properties then, because these particles can only come in integer numbers, different atoms can be combined to form compounds also only in integer numbers. It was an accomplishment of Proust that he was able to correctly distinguish between pure compounds and mixtures.ĭalton argued that atoms could explain the law of the multiple proportions. So, for the example in the two compounds between carbon and oxygen, in mixtures of the two there could be carbon to oxygen ratios varying continuously between 27.7 % to 42.9% carbon, and 42.9% to 72.7% oxygen. When two pure compounds of two different elements are mixed, then the mixture can have a continuous mass ratio between the two elements because compounds can be mixed at any ratio. The very existence of this debate was because at the time the distinction between pure chemical compounds and mixtures had not yet been fully developed. The law was questioned by Proust's fellow Frenchman Claude Louis Berthollet, who argued that the elements could combine in any proportion. Figure 1.1.5 Joseph Proust (1754-1826) (Attribution: The original uploader was HappyApple at English Wikipedia. For example the element analysis of pure substances containing the elements carbon and oxygen would be either 42.9% carbon and 57.1% oxygen or 27.3% carbon and 72.7% oxygen. It was found that a pure substance always contains exactly the same proportion of elements by mass. 
This law was the result of chemical analysis that determined the mass ratio of elements in pure substances. The discovery of the law of the conservation of mass led to the discovery of the law of the constant composition (also called law of definitive proportions) by Joseph Proust (Fig. This implies that for any chemical process in a closed system, the mass of the reactants must equal the mass of the products. An equivalent statement is that matter cannot be created or destroyed, although it may change form. The law of conservation of mass states that the mass of a closed system of substances will remain constant, regardless of the processes acting inside the system. This in turn led to the idea of chemical elements, as well as the idea that all chemical processes and transformations are simple reactions between these elements. When scientists realized that mass never disappeared they could for the first time embark on quantitative studies of the transformations of substances. However, the mass of the wood actually does not disappear it is just converted into the mass of gases, mainly carbon dioxide. For example, the mass of wood seems to disappear when it is burned. Until then, in many instances mass seemed to appear or disappear. Historically, the conservation of mass and weight was kept obscure for millennia by the buoyant effect of the Earth's atmosphere on the weight of gases, an effect not understood until the vacuum pump first allowed the effective weighing of gases using scales. What lead to Dalton’s atom hypothesis? The first discovery that was important to the development of modern atomic theory was the law of the conservation of mass by Antoine Lavoisier (Fig. , )Īlthough the atomistic idea was already known in the antique, it became forgotten for a long time and was only reintroduced about 2000 years later with Dalton’s atom hypothesis. (Attribution: Louis Jean Désiré Delaistre (1800–1871) drawing by Julien Léopold Boilly (1796–1874). Modern Atomic Theories Figure 1.1.4 Antoine Lavoisier, the father of modern chemistry, discovered the law of conservation of mass in 1774. This question would not be answered until about 2000 years later. However, Leukippes and Democrites did not know how many different elements there were, and how different atoms of different elements were actually distinguished. Atomistic element theory allows for many different elements which helps to explain the complexity of the material world satisfactorily.  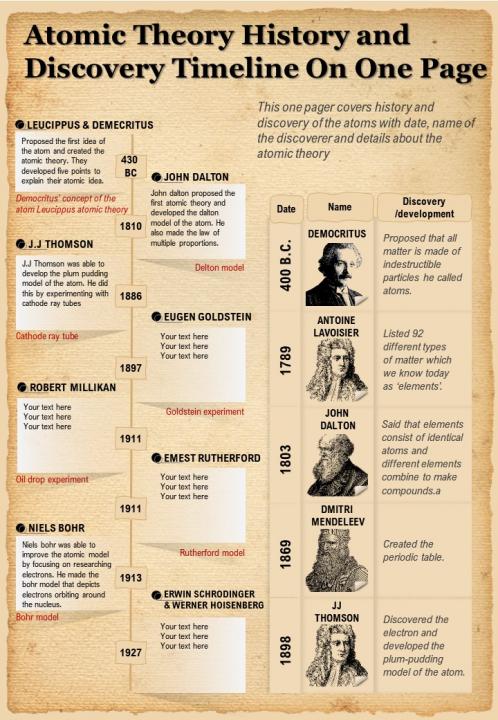
Ultimately, after many divisions, one would arrive at particles that could not be further divided, and these particles would be called atoms. They assumed that particles cannot be divided into smaller particles infinitely often. The first greek philosophers that introduced atomistic element theory were Leukippes, and Democrites (Figure 1.1.3). This means that they did not include the idea that elements were made of small particles that were indivisible. The element theories of Thales, Anaximenes, Heraklitus, and Empedocles were all non-atomistic. \)įirst atomistic ideas Figure 1.1.3 Democrites (460 – 370 BC) (Attribution: Strannik 92 ) 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
